What Is Yield Strength?
July 1, 2009Comments
The initial or elastic portion of a tensile test evaluates the bonding strength between atoms in the metallic unit cell. These extremely strong elastic stresses are balanced to provide a lowest energy state. When an external force is applied to deform the material, the atomic spacing increases (tension) or decreases (compression) causing an increase in the elastic stress. The strength of these elastic stresses is measured by the Young’s Modulus or slope of the curve in the graph of engineering stress versus strain (Fig. 1).
This elastic stress exists only as long as the external force is applied or some of the stresses are trapped by the part geometry. As discussed in the last two columns, these elastic stresses are the source of springback. A simple model replaces the multiplicity of atomic bonds with springs.
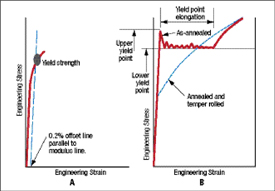
Fig. 1—Schematic A shows yield strength as a smooth transition from elastic to plastic strain. Schematic B shows sharp upper and lower yield points due to yield point elongation.
The greater the applied force, the more the atomic spacing changes and the elastic stress increases. At some point, however, the slope of the curve rapidly decreases. The smooth change in Fig. 1A is called the yield strength, determined by the intersection of the curve and a parallel line offset from the modulus line by 0.2 percent strain. The major component of the deformation now is plastic or permanent deformation (strain). The metallurgical explanation of this plastic deformation is extremely complex. However, there is a simple analogy that will convey sufficient understanding to explain the many s that yield strength can be increased or decreased. The same analogy will be useful in next month’s column dealing with work hardening and the n-value.
In the elastic portion of the deformation, the sheetmetal acted like a perfect arrangement of unit cells throughout the entire sheet. Fortunately for the world, this perfect arrangement does not exist. The sheetmetal is full of discontinuities or gaps at which a line of atoms is missing. Under sufficient external force, atoms on one side of the gap will jump from one side to the other, causing the line of missing atoms to move through the sheet. These moving gaps are called slip lines.
A rug analogy can illustrate how these slip lines create permanent deformation. Assume a huge rug in a room with a very rough floor needs to be moved one foot to the left (Fig. 2). One could pull and pull on the left edge (A) in an attempt to move the entire rug. Probably some of the threads of the rug will elongate elastically. Stop pulling on the rug and the threads will return to their original length (springback). The easy to move the rug is to create a hump of rug one foot in width along the right edge of the rug (B). One can very easily push the hump (a slip line)
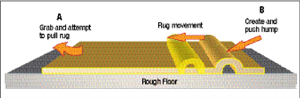 |
| Fig. 2—Schematic showing (A) almost impossible task to move a very large rug to the left by pulling, and (B) an easy move by creating and pushing a hump along the rug. |
Sheet steel coming out of the annealing furnaces has nitrogen atoms “pinning” the slip lines. This requires an initial higher stress to initiate the plastic deformation observed at the upper yield stress in Fig. 1B. In our analogy, these nitrogen atoms are small nails tacking the rug to the floor. A higher force (upper yield strength) is required to get the hump rolling. Once moving, the hump must continue to pull out additional nails as it moves across the room. This creates the almost constant load as the hump (now called a Lüder’s line) moves down the tensile sample. When the steel is temper passed after annealing, the effect of these nails is neutralized. In the old days of rimmed steel, the steel would age over time and the yield point elongation would return as the nitrogen atoms renailed the rug. The solution was to produce steel killed with aluminum. Now the nitride nails are tied to the aluminum and once pulled out are too big to be renailed. The aluminum-killed steels used today do not age.
How are different yield strengths created in steels? Common techniques include thermal-mechanical strengthening. An atom of iron in the unit cell is replaced by an atom of another element that is either larger or smaller than the iron atom. This causes an expansion or contraction within the unit cell that makes it more difficult for slip lines to start moving. Atoms of other metals are placed as extra atoms inside the unit cells as further strengtheners.
Zones of unit cells with identical orientations are called grains. The grain boundaries are stronger than the grain interior. Therefore, smaller grains are stronger than larger grains. Cold working the steel, significantly increasing the amount of carbon, quenching certain microstructures, and other processprocesses can be used to strengthen steel, but the stretchability of the resultant product will be degraded compared to thermal- mechanical strengthening. In contrast, to create a lower strength steel, such as vacuum-degassed interstitial-free steel, the carbon content is greatly reduced, the steel is vacuum-degassed, and the effects of other elements present in the steel are minimized by combining them with titanium, columbium, and other additives.
All the different metal industries have conducted extensive research over many decades to understand how the chemistry and physics of the metals create different properties, experiment with appropriate processing, and to conduct consistency trials. The goal is develop products in a wide range of yield strengths to meet the ever-growing demands of the customer, who requires metal alloys with specific properties. MF
View Glossary of Metalforming Terms
Technologies: Quality Control
Comments
Must be logged in to post a comment. Sign in or Create an Account
There are no comments posted. Quality Control
Quality ControlAmrol Jr. New Starrett President and CEO, Other Executives N...
Wednesday, March 5, 2025
Ascential Technologies Appoints Divisional CEO to Specialty ...
Wednesday, April 24, 2024
 Materials
MaterialsBrinell, Rockwell and Vickers Hardness Testing: Use and Misu...
Daniel Schaeffler Friday, April 1, 2022
 Quality Control
Quality ControlTroubleshooting Sheet Metal Forming Problems, Part 2: The St...
Daniel Schaeffler Friday, February 26, 2021






